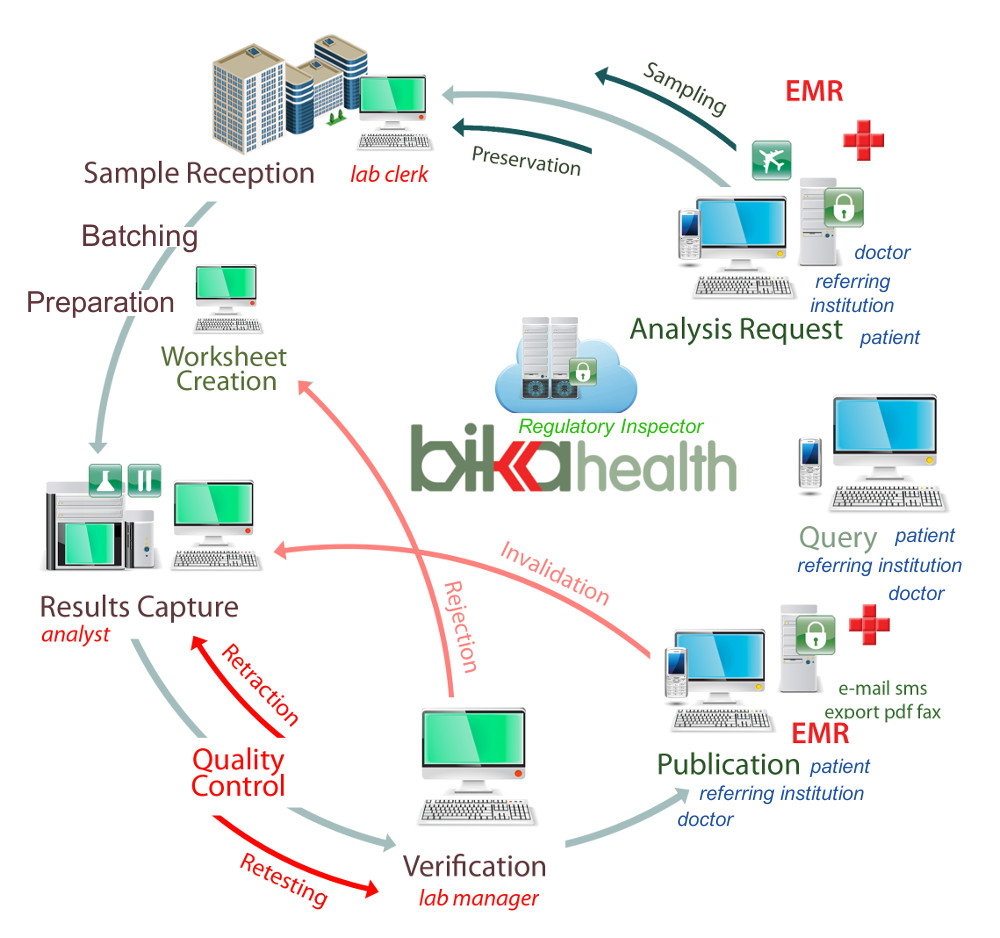
For attendees who complete the class, we also provide access to an online copy of the book. We provide a hardcopy of the training manual. This course is designed for the key individuals who will be involved in the implementation, validation, post-implementation support, and day-to-day use of the Watson LIMS Application. Sample management software your lab will love.Upon completion of this course delegates will have: When registering please note your preference of attendance: Virtual or In-person. Please note that in 2023 this course may be attended in a virtual mode or in person mode in North America. A certified technical training program including instrument integration. Each participant receives a comprehensive training manual that may also be used as a post-training reference. A look at SampleManager 12.0 - The worlds most widely deployed LIMS is now. The software provides a complete audit trail and establishes sample chain of custody. The class format is a mix of lecture and hands-on exercises, including a final practical exercise. Watson LIMS software follows your bioanalytical study from initiation through study closeout, all while enabling compliance with regulations including GLP, 21 CFR Part 11, as well as FDA Bioanalytical Method Validation and EMA guidance documents. and training requirements of the techs, so the lab manager cant assign a. Make Every Sample Matter, Sample Management. This course is delivered in a classroom style by one of our expert education consultants. Pricing, quoting and invoicing can be managed from within SampleManager LIMS. 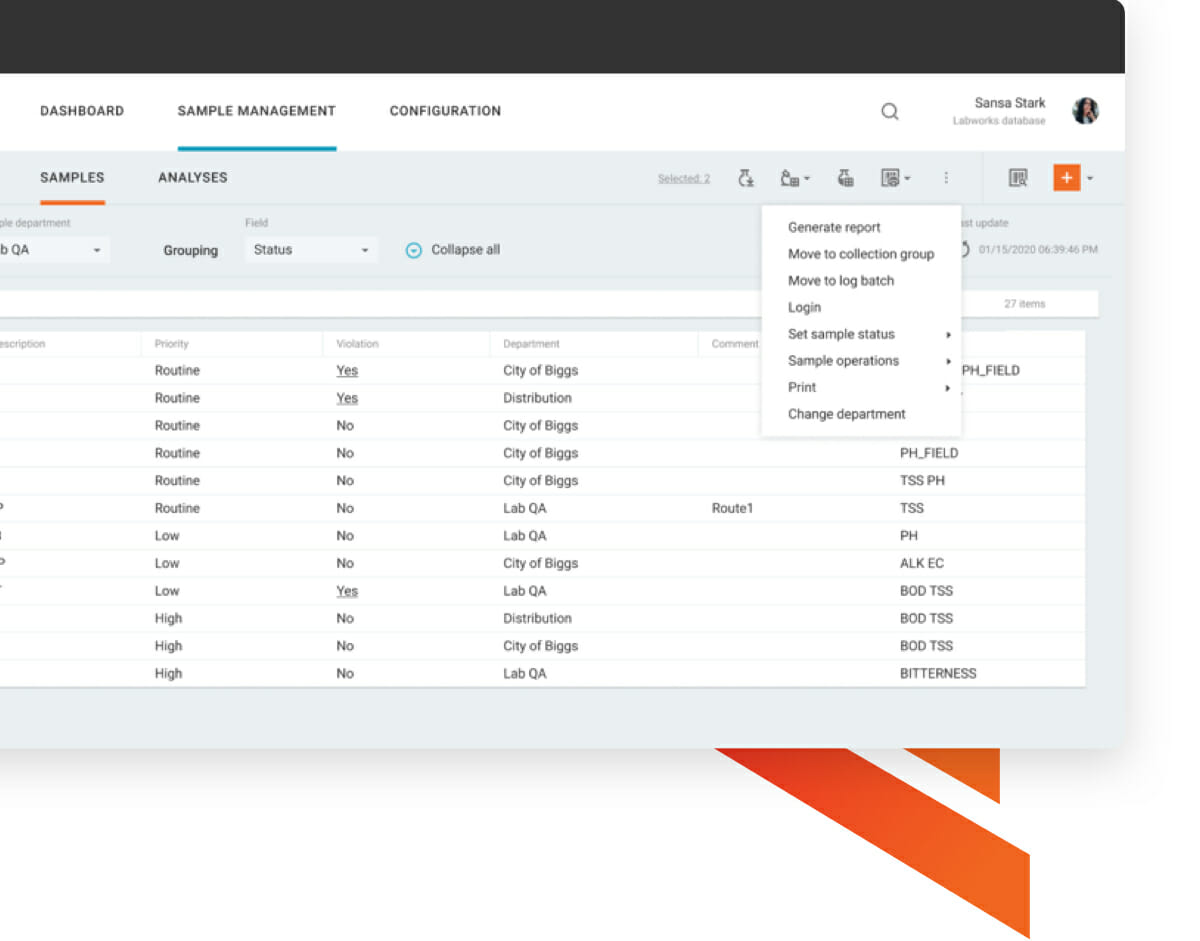
In this three day course participants will acquire the fundamental knowledge needed to configure and use the Watson LIMS product.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
